After covering molecular structures, lipids, and proteins it is time for the sweet stuff. Carbohydrates include sugars, starches, and fiber. Plants generate simple sugars through photosythesis which they use for energy. Plants also use carbohydrates such as cellulose for their structural support, and starch to store energy as sugars in long chains.
We humans use carbohydrates for fast energy. Lipids are the most efficient at energy storage, but carbohydrates are easier to break down into ATP (what our bodies use for energy). We are encouraged to consume complex carbohydrates found in high quantities in grains, potatoes and other starchy foods. Dietary fiber is another important part of a healthy diet which you can get from fruits and vegetables. Simple sugars are just plain fun to eat.
Building Blocks: Monosaccharides
Monosaccharides, or simple sugars, are molecules with one sugar unit. The name is quite a mouthful but it comes from the Latin words mono meaning single and saccharum meaning sugar. All monosaccharides are sweet, but to varying degrees. Three common monosaccharides in food are glucose, fructose, and galactose. Each has the chemical formula C6H12O6.

Monosaccharides have three to seven carbons. The chains can be linear, but a simple sugar with five or six carbons is typically in a ring. There are many different conventions for depicting these sugar rings. The easiest one to draw is a Fischer projection, I think a Haworth projection is the easiest to understand, and the most accurate one is a chair configuration, also known as a spatial projection. The schematics look so different from one another, it is hard to believe they represent the same thing. But they do and the movie below shows how.
Technical Side Note:
The glucose in the video was beta-D-glucose. What is does the beta refer to? It indicates that the OH group of the anomeric carbon (the carbon that is the stereogenic center of the sugar ring) extends up from the carbon. Alpha-glucose would have that OH group drawn down. Whether a monosaccharide is alpha or beta is important because it determines what kind of bond that monosaccharide can form.
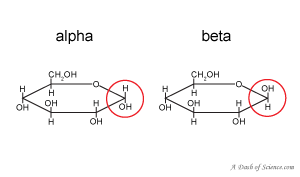
All naturally occurring sugars are D-sugars. D-sugars have a specific type of stereochemistry (spacial arrangement of atoms) that differs from L-sugars. “A D-sugar has the OH group on the stereogenic center farthest from the carbonyl on the right in a Fischer projection.” (Organic Chemistry, 2nd edition, Janice Gorzynski Smith, page 1032)
Disaccharides
Disaccharides also fall under the category of simple sugars. A disaccharide is formed when two monosaccharides bond. The bond is specifically called a glycosidic linkage. The three most common disaccharides are sucrose, lactose, and maltose.
| Disaccharide | Components | Found In |
|---|---|---|
| Sucrose | glucose + fructose | table sugar |
| Lactose | glucose + galactose | milk |
| Maltose | glucose + glucose | malt |
You’ve got to love sucrose, also known as sugar. Cookies, cakes, and candies just would not be the same without it. It comes from sugar canes or sugar beets.
Lactose is well known because some people are lactose intolerant. These people do not have enough lactase, an enzyme that breaks the glycosidic linkage of lactose. The body can absorb galactose and glucose but not lactose. So when a person who is lactose intolerant eats a lot of lactose rich food, they experience gastric distress.

Oligosaccharides
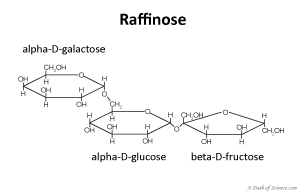
Oligosaccharides are carbohydrates that contain three to ten monosaccharides. Both oligosaccharides and polysaccharides are called complex carbohydrates. Common food oligosaccharides are raffinose and stachyose. They are both found in beans. Our bodies can’t break the bonds between the monosacchride components because of how they are linked but some bacteria in our gut can. The unpleasant side effect is flatulence (excess gas which leads to farting).
Polysaccharides
Three or more monosaccharides make a polysaccharide. Polysaccharides can contain thousands on monosaccharides. Starch and fiber are both examples of polysaccharides.
Starch has two different components: amylose and amylopectin. In spite of having pectin in its name, amylopectin has nothing to do with pectin. Amylose is made of straight glucose chains and amylopectin has branched glucose chains.
Fiber includes any polysaccharide that the human body cannot break down because it lacks the right enzymes. For example, cellulose is a chain of glucose, just like starch. But starch is made of alpha-D-glucose and cellulose is made of beta-D-glucose. That single switch between alpha- and beta- glucose makes such a big difference!
Wrap Up
Carbohydrates are a great source of energy. Every carbohydrate is made up of monosaccharides, which are sweet tasting carbon rings. Along with monosaccharides there are disaccharides, oligosaccharides and polysaccharides.
And that concludes the macro nutrients series!
Sources:
- Organic Chemistry, 2nd edition, by Janice Gorzynski Smith
- Principles of Biochemistry, 5th Edition, by David L. Nelson and Michael M. Cox


3 thoughts on “Macro Nutrients Part 4: Carbohydrates”