Nutrients are split up into two main categories: macro nutrients and micro nutrients. Micro nutrients include vitamins and minerals. They are nutrients that your body needs in small amounts.
Macro nutrients make up the bulk of your diet and provide energy. This category includes lipids, proteins, and carbohydrates. Knowing about each type of macro nutrient helps in understanding why food does what it does.
Since this blog is all about understanding food, some background information about lipids, proteins, and carbohydrates is needed. So, the next few blog posts will focus on the macro nutrients. I’ll go over their chemical structure, basic components, and some common terms. It is not the most exciting stuff in the world, but it is a great foundation for more interesting topics.
Understanding Images of Molecular Structure
I think that images are a great teaching tool. The macro nutrient series will include several illustrations. Just so we are on the same page, the rest of this post is a brief tutorial on how to understand representations of molecular structures.
Kids often draw stick figures. Stick figure people, stick figure pets, and stick figure houses do a great job of representing things. Without shading, different colors, or even clothes on their subjects a child can draw a picture that is easy to understand.
Scientists do the same thing. They have very simple drawings to convey more complex subject matter. For molecules, the stick figure drawings are called skeletal formulas. Skeletal formulas are great because they are so uncluttered. It is really easy to see key structural elements in these minimalistic designs. Even though the drawings are simple, they need some explanation.
How to Interpret Skeletal Formulas
- Letters represent elements from the periodic table.
- A line is a chemical bond.
- If a line juts out into nowhere, assume that there is a carbon atom on the end of that line.
- If two lines intersect but there is no letter, assume that there is a carbon at the intersection.
- Sometimes the hydrogen atoms are left out of the drawing.


3D Perspective
Skeletal formulas are limited because they only show things in 2D. There are a few methods for depicting 3D concepts while still using stick drawings, but in this blog if the 3D perspective is important I will use a different technique.
When representing elements and molecular structures in 3D, the most common method depicts the elements as spheres. Technically, there are electrons zooming around a nucleus in different orbitals but that is a bit complex. So elements are shown as spheres. They differ in size based on how big their nucleus and electron clouds are. Images normally have different colors for each element so it is easier to tell which element is which. Some images include the bonds between elements (the stick between the spheres). Other images depict bonds by having spheres touch.
Of Note
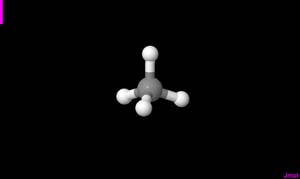
Carbon has four open electrons, which means it can form four covalent bonds. In skeletal formulas, those bonds might be portrayed coming out from the C directly above, below, and out each side. But in the physical world carbon is 3D, not flat. So these bonds are not as easy to depict. If carbon is bonded at all four bonding sites it makes a triangle pyramid (a pyramid with a triangle base instead of a square one). Carbon is at the center of the pyramid and another element sits at every corner of the pyramid.
Summary
Nutrients are divided into macro and micro nutrients. The macro nutrients are lipids, proteins, and carbohydrates. As the main components of our diet they provide the bulk of the energy we get from food. The next few posts will look at each macro nutrient individually.
Chemical structures are portrayed in 2D with skeletal formulas. There are more complex 3D representations as well. Knowing how to interpret these images will be helpful for the next few blog posts:
Photos: skeletal image by Benjah-bmm27; 3D models created with JMol


2 thoughts on “Macro Nutrients Part One: Molecular Structures”